News
SDSC’s Expanse Charts the Hidden Toll of Sickle Cell Disease
Published March 16, 2026
By Kimberly Mann Bruch and Scott Paton
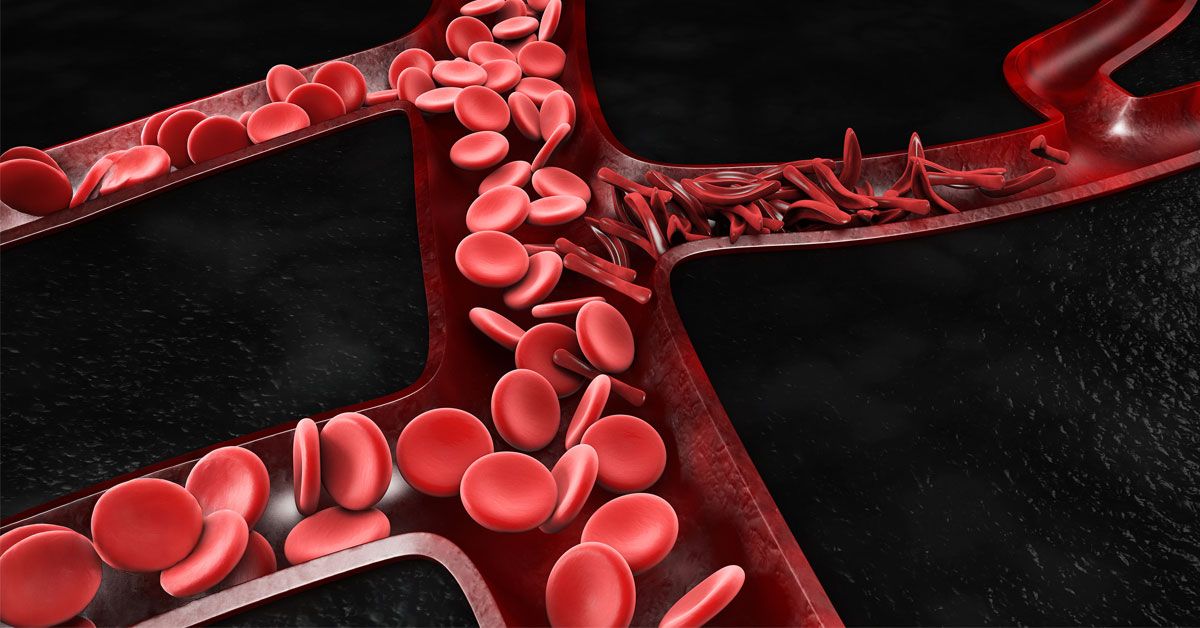
Blood is often pictured as a smooth, continuous stream, but in reality it behaves more like rush-hour traffic in a dense city with crowded lanes, sudden merges and countless tiny intersections where direction and speed change instantly. For most people, those arterial roadways manage this traffic gracefully.
For people living with sickle cell disease (SCD), however, the circulation is under constant strain. Much of the harm happens quietly in the body’s smallest blood vessels, where red blood cells must bend, slide and divide into branching pathways to keep tissues alive. When this choreography breaks down, damage accumulates over years, injuring vessels, stressing organs and shortening lives.
Recent research, supported by U.S. National Science Foundation ACCESS allocations on the Expanse system at the San Diego Supercomputer Center (SDSC), part of the University of California San Diego School of Computing, Information and Data Sciences, zooms in on what happens when blood encounters microscopic vessel bifurcations, the tiniest forks in the road where one capillary splits into two branches.
“Blood isn’t just a liquid, it’s a suspension of living, flexible particles,” said Michael Graham, the Steenbock Professor of Engineering and Vilas Distinguished Achievement Professor of Chemical and Biological Engineering at the University of Wisconsin–Madison. “Once you recognize that, you realize that where cells are located within the flow can matter just as much as how fast the blood is moving.”

Using high-performance computing (HPC) systems like Expanse at SDSC, Graham’s team along with Wilbur Lam, a physician-scientist at Emory University and Georgia Institute of Technology, have shown that in SCD, a subset of abnormal red blood cells consistently takes the wrong turn at these junctions, triggering a chain of mechanical events that may help explain how vessels are gradually injured over time. Graham said that in healthy microcirculation, red blood cells tend to drift toward the center of a vessel as they move along, so when a vessel divides, these centrally located cells are more likely to follow the branch carrying the larger fraction of blood. This pattern helps oxygen distribute efficiently throughout the body’s web of capillaries.
SCD disrupts that balance. Some red blood cells become stiffer, smaller and less flexible with properties that push them away from the center of the vessel and toward the wall, where they travel in a thin layer rather than mixing evenly with the main flow. That seemingly subtle shift has major consequences at a fork in the road.
Simulations supported by NSF ACCESS and run on SDSC’s Expanse showed that when a vessel splits, stiff, sickle-shaped cells near the wall are more likely to peel off into the slower-flowing branch rather than following the faster stream. Over time, that pattern causes certain pathways to become disproportionately loaded with abnormal cells, effectively reversing the usual “traffic rule” that steers centrally located cells into higher-flow branches.
For SCD patients, the key issue is not only which branch a cell enters but what happens once it gets there. After the split, sickle cells tend to cluster along the outer walls of the branches, exactly where the flow can generate strong mechanical forces. When stiff cells repeatedly skim along the vessel lining, they create sharp spikes in shear stress, the frictional force the flowing blood exerts on the wall.
Those stress spikes matter because the endothelium, the inner lining of blood vessels, responds to mechanical forces just as it does to chemical signals. Persistent, elevated stress can trigger inflammation, make vessel walls “stickier” and promote further obstruction. Over years, this wear-and-tear may help explain why people with sickle cell disease face higher risks of stroke, organ damage and chronic pain, even in regions where vessels never become completely blocked.
“Simulations on SDSC’s Expanse let us see patterns that would otherwise remain hidden. For people living with sickle cell disease, that kind of insight offers something invaluable: not just better explanations, but the possibility that smarter, gentler treatments may one day change the course of the disease, one tiny vessel at a time.”
— Michael Graham, Steenbock Professor of Engineering and Vilas Distinguished Achievement Professor of Chemical and Biological Engineering at the University of Wisconsin–Madison
“What we’re seeing with simulations on Expanse is a purely physical mechanism that can contribute to vascular injury,” Graham said. “The cells don’t need to stick chemically or form clots. Their stiffness and location alone are enough to increase stress on the vessel wall.”
At the individual level, this work reframes sickle cell disease as not only a problem of oxygen delivery but also a disorder of cumulative vascular injury. Even when blood continues to move, repeated mechanical insults can quietly damage vessels in the brain, lungs, kidneys and other organs. The study also sheds light on why complications often cluster in specific regions of the circulation rather than appearing uniformly throughout the body. Vessel geometry, including branch diameter and angle of divergence, can amplify these effects and create persistent hot spots of stress.
Graham said that the findings suggest new directions for therapy. Instead of focusing solely on preventing outright blockages, future treatments might aim to change how red blood cells distribute themselves within flowing blood. Drugs that soften cells, reduce dehydration or lessen their tendency to migrate toward vessel walls could lower harmful mechanical stress even if overall flow rates stay the same. Microfluidic devices inspired by this work might allow clinicians to test how an individual patient’s blood behaves under different conditions, opening the door to more personalized treatment strategies.
“Our research does not offer an immediate cure, but it provides a clearer map of how microscopic events translate into long-term harm,” Graham said. “With the help of SDSC’s Expanse system, we have connected the repeated wrong turns of sickle cells at the smallest junctions of the circulatory system to the real-world suffering of patients. Our work shows how damage can accumulate silently and why managing sickle cell disease requires more than simply maintaining blood flow.”
Allocations on SDSC’s Expanse were supported by NSF ACCESS (allocation nos. MCB190100 and PHY240144).
What is sickle cell disease?
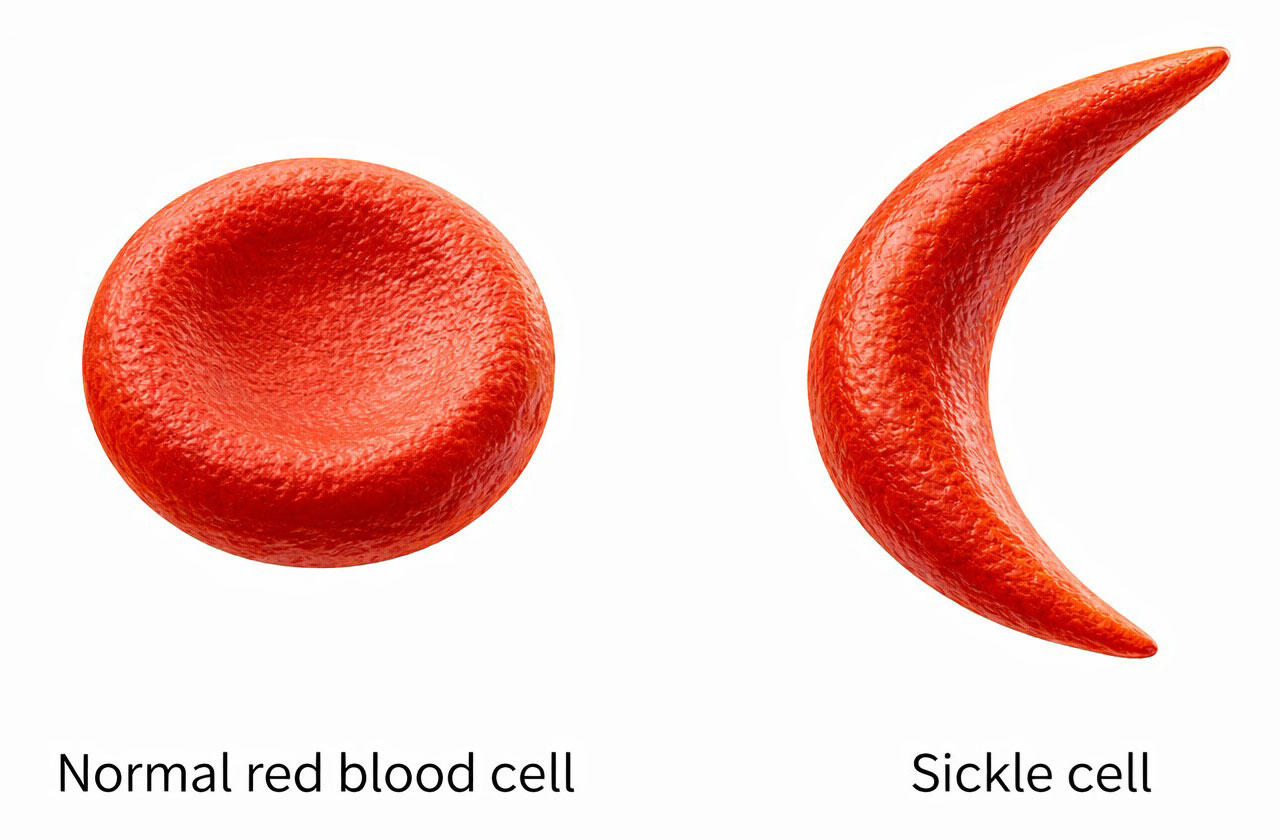
Sickle cell disease (SCD) is an inherited blood disorder that alters the shape and behavior of red blood cells, which normally carry oxygen throughout the body. In SCD, a genetic mutation changes hemoglobin — the oxygen-carrying protein inside red cells — so that under low-oxygen conditions it forms stiff fibers that force cells into rigid, crescent (sickle) shapes instead of remaining soft and round. SCD most commonly affects people of African descent and also occurs in individuals from parts of the Mediterranean, the Middle East, India and Latin America. This pattern reflects evolutionary history rather than any biological hierarchy: carrying a single copy of the sickle cell gene provides partial protection against severe malaria, so in regions where malaria was common, the gene conferred a survival advantage and became more widespread; inheriting two copies, one from each parent, transforms this protective trait into serious disease. Symptoms usually begin in early childhood and can include chronic anemia, episodes of severe pain, frequent infections, delayed growth and fatigue. Over time, repeated vascular damage raises the risk of stroke, kidney failure, lung disease, vision loss and shortened life expectancy. Treatment has improved greatly in recent decades. Current options include pain management, antibiotics, blood transfusions and medications such as hydroxyurea that reduce complications, while bone marrow and stem cell transplants can cure some patients, though suitable donors are rare and the procedures carry risks. Hope is growing as advances in gene therapy, better supportive care and deeper understanding of blood flow and vessel damage, including the research described here, bring safer and more widely accessible cures closer to reality. |